Biology
Mar 21, 2026
Molecular Biology
Genetics
Biochemistry
Cell Biology
Evolutionary Biology
Physiology
How Engineered Probiotics Turn Tumors Into Local Drug Factories
A new mouse study engineered the probiotic E. coli Nissle 1917 to home to tumors and manufacture the cancer drug romidepsin on site. Here’s how it works, why it reduced toxicity, a
The headline idea sounds almost like science fiction: inject a probiotic, let it find a tumor, and have it manufacture a cancer drug exactly where it is needed. But the deeper story is not just that bacteria can be engineered. It is that tumors create a strange ecological niche—low oxygen, dead tissue, leaky vessels, weak local clearance—that some microbes can exploit far better than conventional drugs can. That is the layer that makes this result genuinely important.
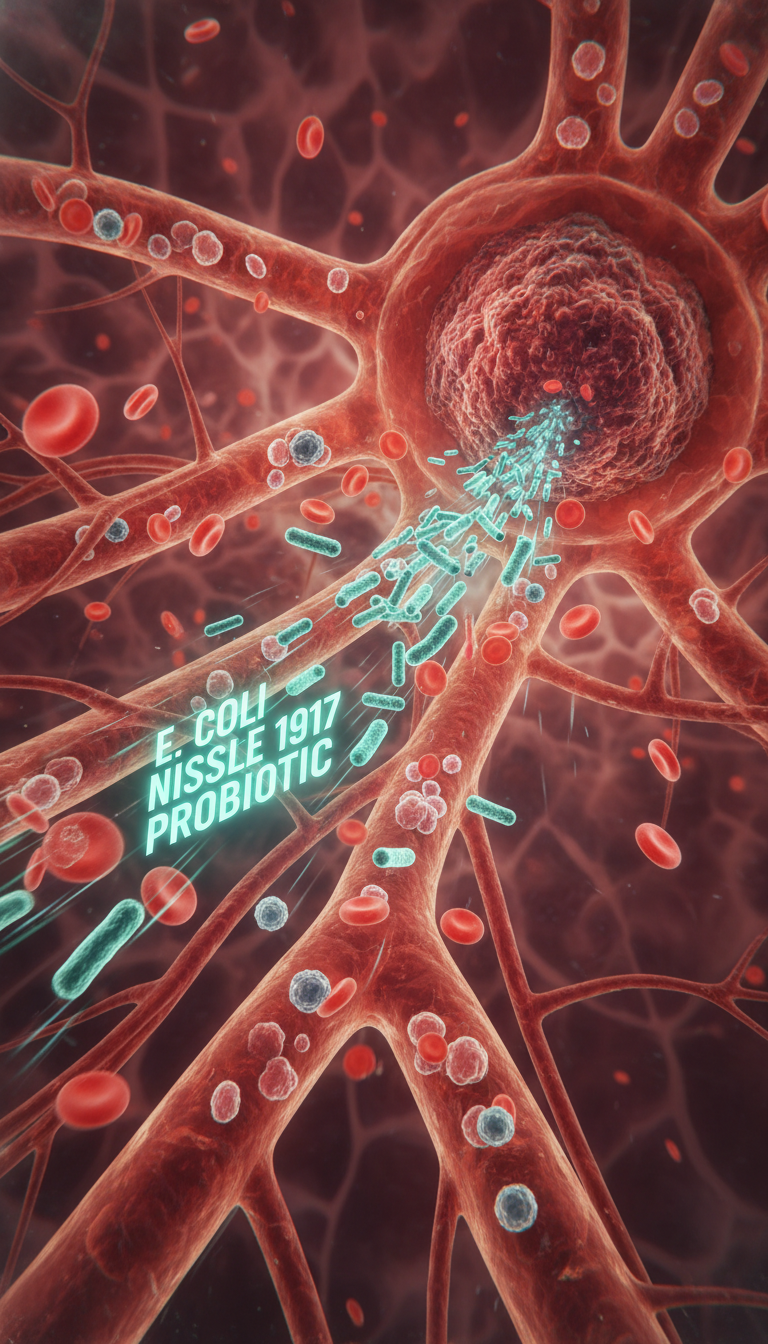
In a March 2026 PLOS Biology study, researchers engineered Escherichia coli Nissle 1917, a well-known probiotic strain called EcN, to produce romidepsin, an FDA-approved anticancer compound. In mouse breast tumors, the bacteria accumulated in tumors, synthesized the drug locally, slowed tumor growth, and avoided the lethal toxicity seen with systemic romidepsin in the same model. That does not mean cancer has been “cured by probiotics.” But it does show why living therapeutics are attracting serious attention.
Why a probiotic can reach tumors in the first place
EcN is not magic, and it is not uniquely “smart.” Tumors simply offer conditions that many normal tissues do not. Their blood vessels are disorganized and leaky, immune surveillance can be patchy, and the core often becomes hypoxic or necrotic. Those regions are hard for many drugs to penetrate but can be hospitable to facultative anaerobic bacteria like EcN.
That is why tumor-targeting bacteria have been explored for years. The appeal of EcN over more aggressive species such as Salmonella is safety: it already has a history as a probiotic and is generally considered less pathogenic. In this study, EcN reportedly accumulated around 100 times more in tumors than in blood or serum, reinforcing the idea that tumors can act as microbial refuges.
What the engineers actually changed
The most impressive part was not merely putting a drug gene into bacteria. Romidepsin is a complex natural product made through a nonribosomal peptide synthetase–polyketide synthase pathway originally associated with Chromobacterium violaceum. Rebuilding that biosynthetic machinery inside EcN required pathway optimization, including genetic tuning to raise output dramatically.
Explore our free biology courses
Molecular Biology
University · Biology
Genetics
University · Biology
Biochemistry
University · Biology
Cell Biology
University · Biology
Evolutionary Biology
University · Biology
Physiology
University · Biology
The team reported a romidepsin yield of about 1.46 mg/L in culture, roughly 27 times the baseline version. In tumors, after induction with arabinose, bacterial production reached up to about 1.5 micrograms per gram of tumor tissue. That matters because the concept only works if the bacteria make enough drug locally to affect cancer cells without flooding the whole body.
A key misconception is that these bacteria autonomously sense a tumor and instantly start producing therapy. In this experiment, drug production still depended on an external inducer. So this is a controllable platform, not yet a fully self-regulating one.
Why local production may reduce toxicity
Romidepsin can be effective, but systemic exposure is limited by side effects, including cardiotoxicity. The logic of the bacterial approach is simple: if the drug is made mostly inside the tumor, the tumor sees a high local concentration while the rest of the body sees less.
That appears to be what made the mouse result striking. Engineered EcN matched or exceeded the antitumor effect of conventional romidepsin treatment, yet the systemic drug group showed mortality while the bacterial-delivery group did not. This is the real promise of “living drug factories”: not just stronger treatment, but a better therapeutic index.

The study also suggested the effect was not purely direct toxicity to tumor cells. Proteomic and molecular analyses indicated broader biological changes, including upregulation of tumor-suppressive pathways such as Ptrh1 and changes in proteins linked to tumor progression. In other words, the bacteria may be reshaping the tumor microenvironment as well as delivering a drug.
What the mouse data does—and does not—prove
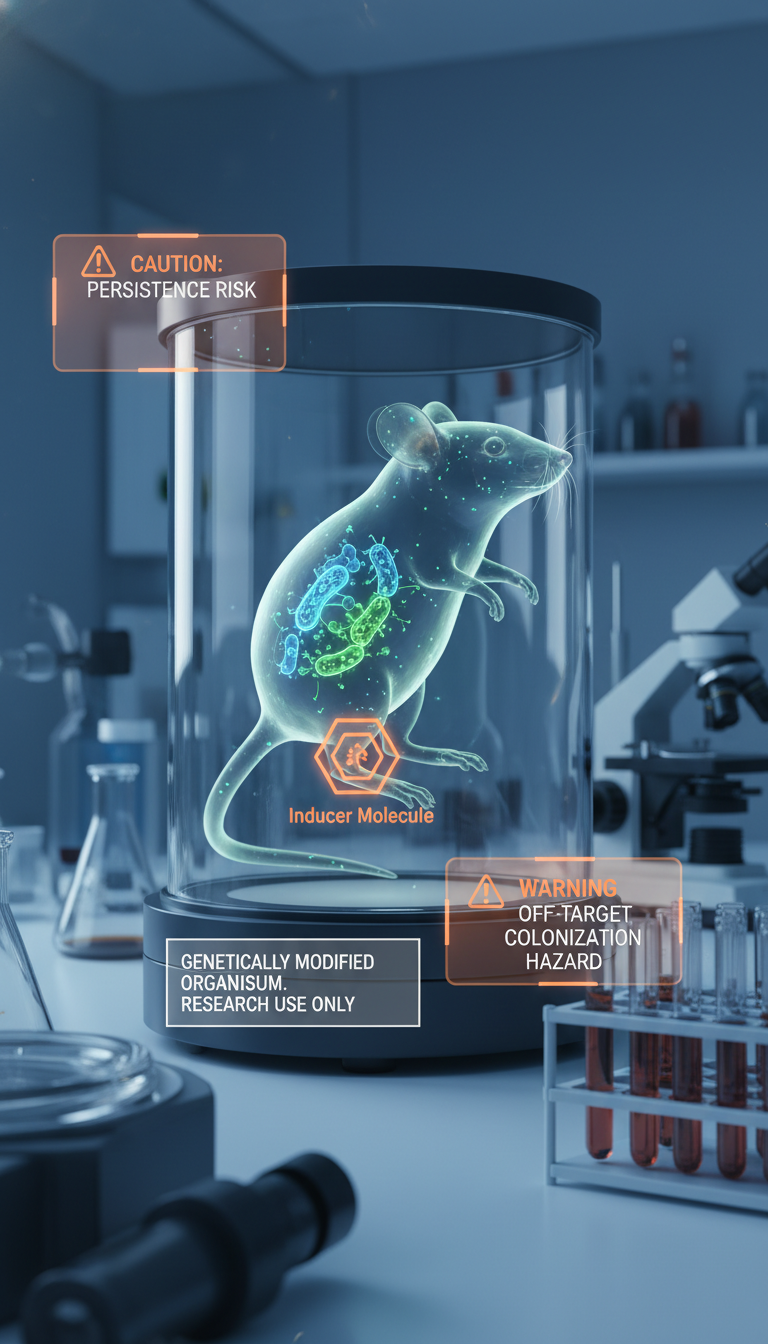
The evidence is promising but preclinical. The work was done in a 4T1 breast cancer mouse model, not in human patients. Mouse tumors are useful, but they do not fully capture the diversity, scale, immune complexity, and treatment history of human cancers.
Several open questions matter:
- Will EcN colonize human tumors as reliably as mouse tumors?
- Can dosing and induction be controlled safely in patients?
- How long do the engineered bacteria persist, and how are they cleared?
- Could off-target colonization, infection, or inflammatory reactions emerge?
- How do regulators evaluate a therapy that is both a microbe and a drug factory?
Those are not minor details. They are the main reason this is exciting science rather than a near-term cure.
What comes next for living cancer therapeutics
The biggest next step is not simply “test in humans.” It is engineering control. Clinically useful strains will likely need kill switches, more reliable genetic stability, better tumor-specific activation, and perhaps combination designs that work with immunotherapy rather than alone.
If those hurdles can be solved, the broader implication is huge: tumors might become manufacturing sites for therapies that are too toxic, too unstable, or too hard to deliver systemically. That could include not only small molecules like romidepsin, but cytokines, immune modulators, or multi-step drug circuits.
This study matters because it moves the field from “bacteria can reach tumors” to “bacteria can make a clinically relevant drug there and change the toxicity tradeoff.” The breakthrough is real. So is the distance to the clinic. The future of cancer therapy may include living medicines—but only if biology, engineering, and safety can be made to work together.