Biology
Mar 23, 2026
Molecular Biology
Genetics
Biochemistry
Cell Biology
Evolutionary Biology
Physiology
A Blood RNA Signal That Predicts Near-Term Survival Better Than Age
Researchers found a small set of blood piRNAs in adults over 70 that predicted two-year survival more accurately than age, lifestyle, cholesterol, physical function, and hundreds o
A new study suggests that a tiny set of blood RNAs may say more about near-term survival in older adults than many familiar health metrics. That is striking on its own. But the deeper story is not just that a biomarker predicts risk. It is why these molecules were surprising candidates in the first place, and why prediction is not the same thing as a ready-made anti-aging therapy.

What the study actually found
In plasma samples from more than 1,200 adults aged 71 and older, researchers identified six piwi-interacting RNAs, or piRNAs, whose lower levels predicted two-year survival with strong accuracy. Reported performance reached roughly AUC 0.86 to 0.92, and the signal held up in an independent validation cohort.
That matters because the piRNA signature outperformed age, lifestyle factors, cholesterol, physical function, and more than 180 other clinical measures. Even more surprising, a simpler model using five piRNAs plus two clinical variables performed about as well as a larger model. In other words, this was not just a weak statistical curiosity buried in a huge dataset.
But an important correction is needed: this does not mean scientists can now predict a person’s total lifespan from a blood draw. The result is about short-term survival risk in people already over 70, not a universal “how long will you live?” test.
Explore our free biology courses
Molecular Biology
University · Biology
Genetics
University · Biology
Biochemistry
University · Biology
Cell Biology
University · Biology
Evolutionary Biology
University · Biology
Physiology
University · Biology
Why piRNAs are such an unexpected signal
piRNAs are best known from the germline, where they help silence transposable elements, sometimes called “jumping genes,” and help maintain genome stability. They have not traditionally been the headline molecules in mainstream aging research, especially not as circulating blood markers.
That is why this finding stands out. If these RNAs are showing up in plasma and tracking survival, they may be reflecting something fundamental about how the body handles stress, damage, inflammation, immune function, or cellular repair in late life. The study also reported that nine piRNAs overall were lower in longer-lived individuals, reinforcing the idea that this is not a one-off signal from a single molecule.
The surprise is not merely technical. It hints that aging biology may be readable through functional regulatory RNAs, not just through standard lab values or broad “aging clocks.”
How could lower piRNA levels influence survival?
The honest answer is that the full mechanism is still unresolved. But there are several biologically plausible paths.
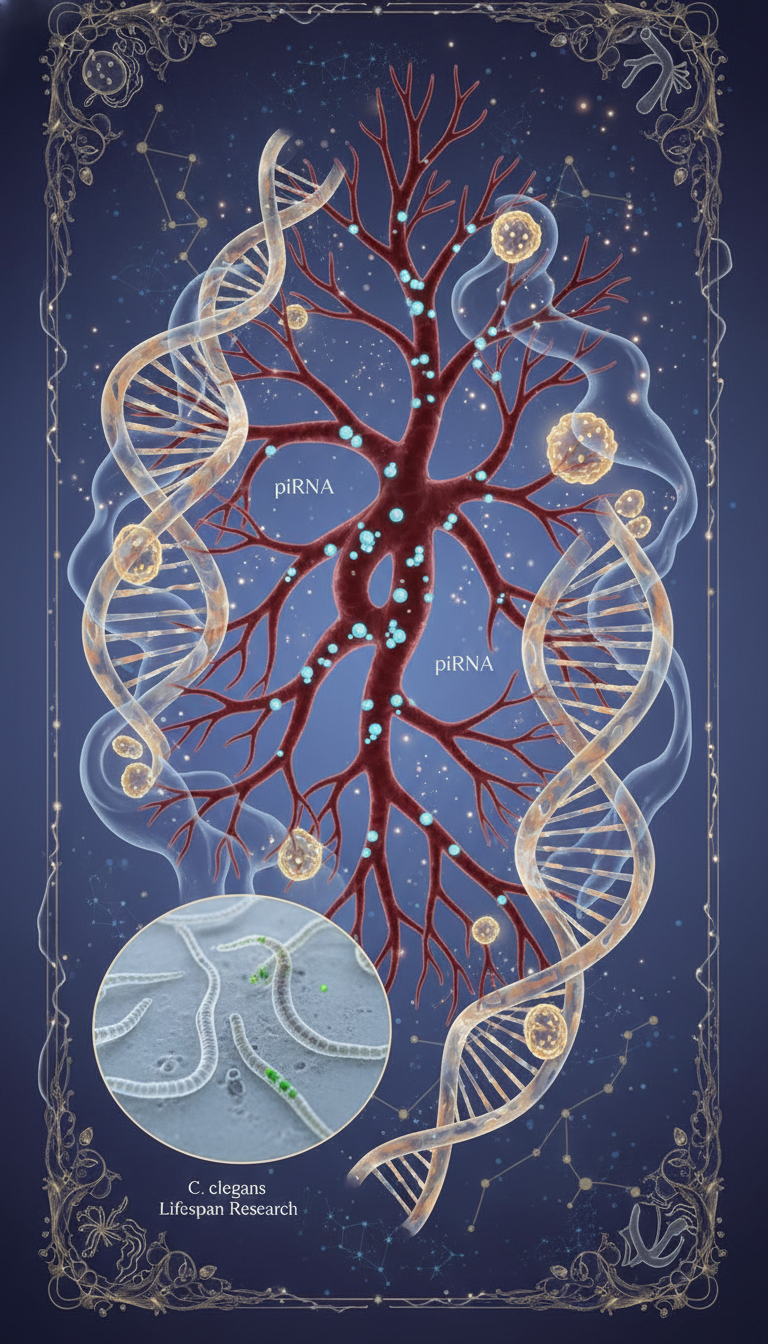
- Genome stability and stress response: piRNAs help regulate transposons and gene expression. In aging tissues, misregulation of these systems could amplify cellular stress.
- Immune and inflammatory signaling: circulating piRNAs may reflect how tissues communicate damage or resilience, especially when the immune system is under chronic age-related strain.
- Tissue-specific release: blood piRNAs may be signals shed from particular organs under stress, making them indirect readouts of systemic decline.
The most provocative clue comes from C. elegans, where reducing piRNA biogenesis reportedly doubled lifespan. That does not prove the same causal pathway exists in humans, but it raises a serious possibility: lower levels may not just mark resilience, they may participate in it.
Still, this is where caution matters. The human study is observational, and even sophisticated causal AI methods cannot replace direct intervention experiments. A biomarker can sit close to a cause without being the cause itself.
Why this is not yet a consumer longevity test
Two things can be true at once: the result is exciting, and it is not ready for commercial overpromising.
First, the cohorts were older adults, with limited diversity reported in the trend summary. We do not yet know whether the same signature works in younger adults, different ancestries, or people with different disease profiles. Second, prediction over two to five years is clinically useful, but very different from forecasting lifetime aging trajectory.
There is also a practical issue: a good biomarker should be reproducible across labs, stable across sample handling conditions, and interpretable alongside medications, infections, cancer, frailty, and other confounders. That work still needs to be done.
What experts will want to know next
The next phase is not just “bigger studies.” It is a chain of harder questions: which tissues produce these circulating piRNAs, what controls their levels, and whether changing them can safely change outcomes.
If future work shows that specific piRNAs actively shape stress resistance, they could become more than biomarkers. They could become targets for precision interventions. But if they mainly reflect hidden tissue damage, their value may be strongest in prognosis and risk stratification rather than therapy.

The bottom line
This study did not discover a magic lifespan meter. It found something arguably more interesting: a small RNA signature in blood that may capture biological resilience in late life better than many standard clinical measures. The two key open questions are how circulating piRNAs influence survival biologically and whether they can be safely targeted rather than merely measured. Until those are answered, the finding is best seen as a powerful clue about aging, not a finished longevity product.